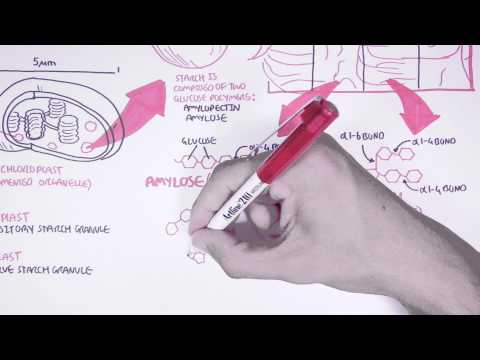
0:00 Carbohydrates are the most abundant biomolecules on Earth. 0:05 They are found everywhere. 0:07 Carbohydrates are a major source of energy for all living organisms, such as 0:11 animals 0:11 and plants. 0:14 But they are not only important for energy. 0:17 Carbohydrates also serve as important structural components. 0:22 For example, DNA contain the carbohydrate ribose, and the plant cell wool are 0:28 made up of the 0:29 carbohydrate cellulose. 0:32 Carbohydrates mainly contain carbon, hydrogen, and oxygen atoms. 0:37 In a molar ratio of one to one, one carbon, two hydrogens, and one oxygen. 0:47 Carbohydrates can be divided into four types. 0:49 These are monosaccharides, disaccharides, oligosaccharides, and polysaccharides 0:56 . 0:56 The word saccharide is derived from the Greek word for sugar. 1:01 Now let us look at each of these types of carbohydrates and learn a bit more 1:06 about their 1:06 structure and how they are formed. 1:10 This is a biochemistry lesson. 1:15 Let's begin with monosaccharides. 1:18 Monosaccharides are also referred to as simple sugars, and they are the 1:21 smallest units that 1:22 make up any carbohydrate. 1:25 They are the building blocks. 1:28 The three main monosaccharides in the human diet include glucose, galactose, 1:34 and fructose. 1:35 Now these structures may look intimidating, but all you need to know is that 1:38 they contain 1:39 carbons, hydrogens, and oxygens. 1:44 Let's look at glucose first, which you all probably have heard of. 1:48 Glucose is the main source of energy for humans. 1:53 Here you can see glucose in its cyclic chemical form. 1:56 What's important to know about glucose is that it contains six carbon atoms, so 2:00 let's 2:00 have a closer look. 2:04 Here C represents a carbon atom, and we can label these carbon atoms of glucose 2:09 with numbers 2:10 1, 2, 3, 4, 5, 6 in this specific order. 2:16 So glucose has six carbon atoms. 2:21 This particular type of glucose is actually an alpha glucose because it has an 2:28 alpha configuration. 2:33 Alpha carbohydrates is where the hydroxyl group, the OH group of carbon number 2:39 1, is pointing 2:40 in the opposite direction to the carbon number 6, so these are opposite each 2:49 other. 2:51 And there is another type of glucose known as a beta glucose. 2:54 This is essentially where the hydroxyl group of carbon number 1 and carbon 2:59 number 6 are 3:00 pointing in the same direction. 3:03 So beta carbohydrates is where the hydroxyl group of carbon number 1 is 3:09 pointing in the 3:10 same direction as carbon number 6. 3:16 And again, these alpha and beta carbohydrates, they also apply to other types 3:23 of carbohydrates 3:25 such as galactose as well as fructose. 3:29 So for example, this galactose molecule is actually a beta galactose because 3:34 the hydroxyl 3:35 group here and the carbon number 6 is pointing in the same direction. 3:40 Similarly, this fructose here is actually in a beta configuration, so it's a 3:46 beta fructose 3:47 because the hydroxyl group here is pointing in the same direction as carbon 3:55 number 6. 3:57 So I hope you understood the structure of the three major monosaccharides in 4:02 the human 4:03 diet. 4:06 Now let's look at disaccharides. 4:09 Monosaccharides are made up of two monosaccharides. 4:13 So for example, a glucose molecule and another glucose molecule can form a bond 4:19 with each 4:20 other. 4:22 This new disaccharide is called maltose. 4:26 Maltose is essentially two glucose molecules linked together. 4:31 It is linked together by an alpha 1 to 4 glycosidic bond. 4:35 It's called alpha 1 to 4 glycosidic bond because carbon number 1 of this 4:43 glucose and 4:44 carbon number 4 of this glucose are involved in the linking process. 4:49 And it's called alpha because both these glucose molecules are in an alpha 4:57 configuration. 4:58 Now the process of linking monosaccharides with one another is called cond 5:03 ensation. 5:04 And here, water is released. 5:07 Therefore the reverse reaction is hydrolysis and this is where we add water. 5:15 Adding water to an alpha 1 to 4 glycosidic bond will break the bond. 5:22 So maltose is only one example of a disaccharide. 5:26 Let's look at some other common examples. 5:29 Now this galactose molecule can link with a glucose molecule. 5:33 So this particular glucose molecule is actually in a beta configuration because 5:38 remember the 5:39 hydroxyl group here is in pointing the same direction as carbon number 6. 5:44 So this galactose molecule and the glucose molecule can form a link and through 5:49 the condensation 5:50 process it will form lactose. 5:54 Lactose is made up of galactose and glucose. 6:00 The galactose and glucose is linked together by a beta 1 to 4 glycosidic bond. 6:06 Now it is called a beta 1 to 4 glycosidic bond because the galactose and 6:11 glucose are 6:12 in a beta configuration and also carbon number 1 and carbon number 4 are 6:18 involved. 6:19 The reverse reaction to break lactose requires hydrolysis by adding water. 6:28 Now lactose as you will probably know is found naturally in milk. 6:36 The third type of disaccharide I want to talk about is where we form a bond 6:40 between 6:41 one glucose molecule and one fructose. 6:44 This glucose molecule is an alpha glucose because as you can see the hydroxyl 6:51 and carbon 6:52 number 6 are pointing the opposite direction. 6:56 So glucose and fructose can form a link and through the condensation reaction 7:02 removal 7:02 of water it can form a disaccharide called sucrose. 7:11 Sucrose is made up of one glucose and one fructose. 7:16 The bond between the glucose and fructose is a little more complicated as it is 7:20 a glucose 7:21 alpha 1 and fructose beta 2 bond. 7:26 This sort of linking occurs. 7:28 So the glucose is in an alpha configuration and the fructose is in a beta 7:33 configuration 7:34 and it is carbon number 1 of glucose and carbon number 2 of fructose that are 7:38 involved 7:39 in the linking process. 7:45 So essentially fructose flips over. 7:50 Sucrose as you all know is table sugar and is formed by plants and not formed 7:56 by animals. 7:58 So humans cannot form sucrose. 8:04 Sucrose is broken down through a hydrolysis reaction. 8:10 So the disaccharides, maltose, lactose and sucrose are all good examples that 8:14 we encounter 8:15 in our normal diet. 8:18 Now let's look at oligosaccharides. 8:21 Oligosaccharides basically consist of short chains of monosaccharides, 8:27 typically less 8:28 than 20 monosaccharides linked together. 8:32 Actually, a disaccharide can be referred to as an oligosaccharide. 8:40 Now let's look at an example of an oligosaccharide. 8:43 So if we were to take this maltose and add another glucose molecule to it, 8:49 through a 8:50 condensation reaction again, we can form an additional alpha 1 to 4 glycosidic 8:56 bond. 8:57 This oligosaccharide is called maltotriose, triacin 3. 9:03 And this maltotriose is made up of glucose and they are linked together as I 9:07 mentioned 9:08 by alpha 1 to 4 glycosidic bonds. 9:12 To break down these bonds requires hydrolysis, the addition of water. 9:19 And this structure can keep growing with the addition of more glucose molecules 9:24 . 9:24 But when the oligosaccharide eventually exceeds 20 monosaccharides with 20 9:30 bonds, the carbohydrate 9:32 is then referred to as a polysaccharide. 9:35 So from an oligosaccharide, it becomes a polysaccharide. 9:41 Most carbohydrates found in nature occur as polysaccharides. 9:46 Polysaccharides are also known as glycans. 9:49 To simplify things, polysaccharides can be a homopolysaccharide, or they can be 9:54 a heteropolysaccharide. 9:56 A homopolysaccharide means the polysaccharide only contains a single type of 10:02 monosaccharide. 10:03 For example, it only contains glucose molecules linked together. 10:09 A heteropolysaccharide means that the polysaccharide contains two or more 10:14 different monosaccharides. 10:15 So for example, a long chain of fructose and glucose molecules. 10:22 To make things a little bit more interesting, a polysaccharide can also be unbr 10:27 anched like 10:28 what you see here, or it can be branched. 10:31 This goes for both homopolysaccharides as well as heteropolysaccharides. 10:37 Polysaccharides can also be unbranched or branched. 10:45 What you have to understand is that polysaccharides I am currently drawing are 10:49 very small. 10:50 In reality, the polysaccharide contains thousands, are made up of thousands of 10:56 monosaccharides 10:57 linked together. 11:00 In this section of the video, we won't focus on heteropolysaccharides, but we 11:04 will look 11:05 at homopolysaccharides because homopolysaccharides serve as storage forms of 11:11 monosaccharides in 11:12 both humans and plants and even bacteria, so they are very, very important. 11:19 So let's look at some examples of homopolysaccharides. 11:24 Starch is a storage form of monosaccharides in plants. 11:30 Starch is the main carbohydrate in the human diet and are found in our bread, 11:34 cereal and 11:35 rice. 11:38 Starch is only made up of glucose because it is a homopolysaccharide. 11:43 Let us zoom into this area here and learn a bit about the bonds. 11:51 So here we have our regular alpha-124 glycosidic bond between two glucose 11:59 molecules, and this 11:59 is because carbon number one and carbon number four of these glucose molecules 12:03 are involved 12:04 in the linking process. 12:06 However, the branching points here is actually an alpha-126 glycosidic bond 12:12 between two glucose 12:13 molecules, and that is because carbon number one of this glucose and carbon 12:18 number six here 12:18 are involved in the linking process. 12:23 So what to take out of this is that Starch has two forms. 12:26 It can be branched, like what I just explained, or starch can be unbranched. 12:31 If starch is unbranched, so it is only a chain of glucose linked together by 12:37 alpha-124 glycosidic 12:38 bonds, it is referred to as amylose. 12:43 If starch is branched, it contains both alpha-124 and alpha-126 glycosidic 12:49 bonds between glucose, 12:52 and therefore it is referred to as amylopectin. 12:58 Amylose and amylopectin are two forms of glucose polymers. 13:04 The other good example of a polysaccharide is glycogen. 13:09 Now glycogen is a homopolysaccharide because it is made up of glucose. 13:16 Glycogen can also be branched or unbranched. 13:20 Glycogen is a storage form of glucose in animals such as humans. 13:27 Starch and glycogen are actually very similar in structure. 13:31 They both are made up of glucose and they can either be branched or unbranched. 13:37 So both starch and glycogen contain amylose and amylopectin. 13:46 The only difference is that glycogen has these branched points occurring every 13:51 8 to 12 glucose 13:52 residues. 13:54 In starch, these branched points occur every 24 to 30 glucose residues. 14:00 So the branched points occur more frequently in glycogen than starch. 14:05 And this of course will influence the structure in some way. 14:12 Another type of polysaccharide is dextrans. 14:17 Dextrans are structural components in bacteria and yeast. 14:22 These polysaccharides are made up of alpha 1, 2, 3 and alpha 1, 2, 6 glycosidic 14:29 bonds. 14:30 So here we have glucose units with alpha 1, 2, 3 bonds and alpha 1, 2, 6 bonds. 14:36 However, the dextrans can also contain alpha 1, 2 and alpha 1, 2, 4 glycosidic 14:43 bonds. 14:44 Finally, the other polysaccharide worth mentioning is cellulose. 14:49 Cellulose are structural components in plants. 14:52 They make up the plant cell wall. 14:55 They are unbranched homopolysaccharide consisting of thousands of glucose 15:01 molecules. 15:03 So here you can see unbranched cellulose on top of each other. 15:09 In cellulose, the glucose molecules have a beta configuration. 15:15 And therefore, the bonds between these glucose molecules are beta bonds. 15:25 The glucose molecules are linked together by beta 1, 2, 4 glycosidic bonds. 15:32 Humans do not have enzymes that break down that hydrolyze beta 1, 2, 4 glycosid 15:40 ic bonds 15:40 of cellulose. 15:42 And so humans cannot digest cellulose. 15:47 Now even though cellulose are only chains of beta glucose, so unbranched beta 15:54 glucose, 15:55 these chains can form hydrogen bonds with each other, forming a very strong 16:09 structure.