Overview
Goodpasture syndrome, now more precisely termed anti-glomerular basement membrane (anti-GBM) disease, is a rare autoimmune small-vessel vasculitis affecting lungs and kidneys, typically manifesting with diffuse alveolar hemorrhage and rapidly progressive glomerulonephritis.
Microscopic polyangiitis (MPA) has an estimated incidence of approximately 0.5 to 1.8 cases per million per year. It demonstrates a bimodal age distribution, with peaks occurring between 20–30 years and again between 60–70 years. There is a male predominance. Genetic associations include HLA-DR15 and HLA-DR4, and environmental triggers such as smoking and hydrocarbon exposure have also been implicated in disease development.
Definition
Anti-GBM disease: autoimmune condition where antibodies target the α3-NC1 domain of type IV collagen in glomerular and alveolar basement membranes
Pulmonary-renal syndrome: simultaneous involvement of lungs (hemorrhage) and kidneys (glomerulonephritis).
Rapidly progressive glomerulonephritis (RPGN): rapid loss of kidney function often with crescents on biopsy, can be manifestation of anti-GBM disease.
Type II hypersensitivity: immune reaction where antibodies bind antigens on tissues, causing complement fixation and inflammation (mechanism here).
Anatomy and Physiology
Relevant normal anatomy/physiology without disease:
- Glomerular basement membrane (GBM): multilaminar basal lamina between endothelial cells and podocytes—critical filtration barrier in kidney glomerulus
- Alveolar basement membrane: part of the thin blood-air barrier for gas exchange in alveoli, consisting of alveolar epithelial and capillary endothelial layers plus basement membranes
Aetiology and Risk Factors
Aetiology: Autoantibodies directed against the non-collagenous (NC-1) domain of α3 chain of type IV collagen in lung and kidney basement membranes
Risk factors
- Genetic predisposition: HLA-DR15, HLA-DR4, HLA-DRB1*1501/1502
- Environmental triggers: cigarette smoking, hydrocarbon/organic solvent inhalation, viral respiratory infection (e.g., influenza) [1,8,4].
- Others: exposure to certain chemicals, infections, possibly post-renal transplant in Alport syndrome, heavy metals [4,8].
Remember
genetic predisposition creates susceptibility—but environmental “hits” often precipitate disease.
Pathophysiology
- Environmental insult (e.g. smoking) → alveolar capillary injury and increased antigen exposure.
- Autoimmune response: B cells produce anti-GBM antibodies targeting α3-NC1 type IV collagen
- Circulating antibodies bind linear epitopes in GBM and alveolar BM → complement activation → neutrophil-mediated inflammation and capillaritis
- Tissue damage: crescentic glomerulonephritis in kidneys; diffuse alveolar hemorrhage in lungs
Think
early damage to barrier exposes hidden antigens, tipping into autoimmune cascade.
Clinical Manifestations
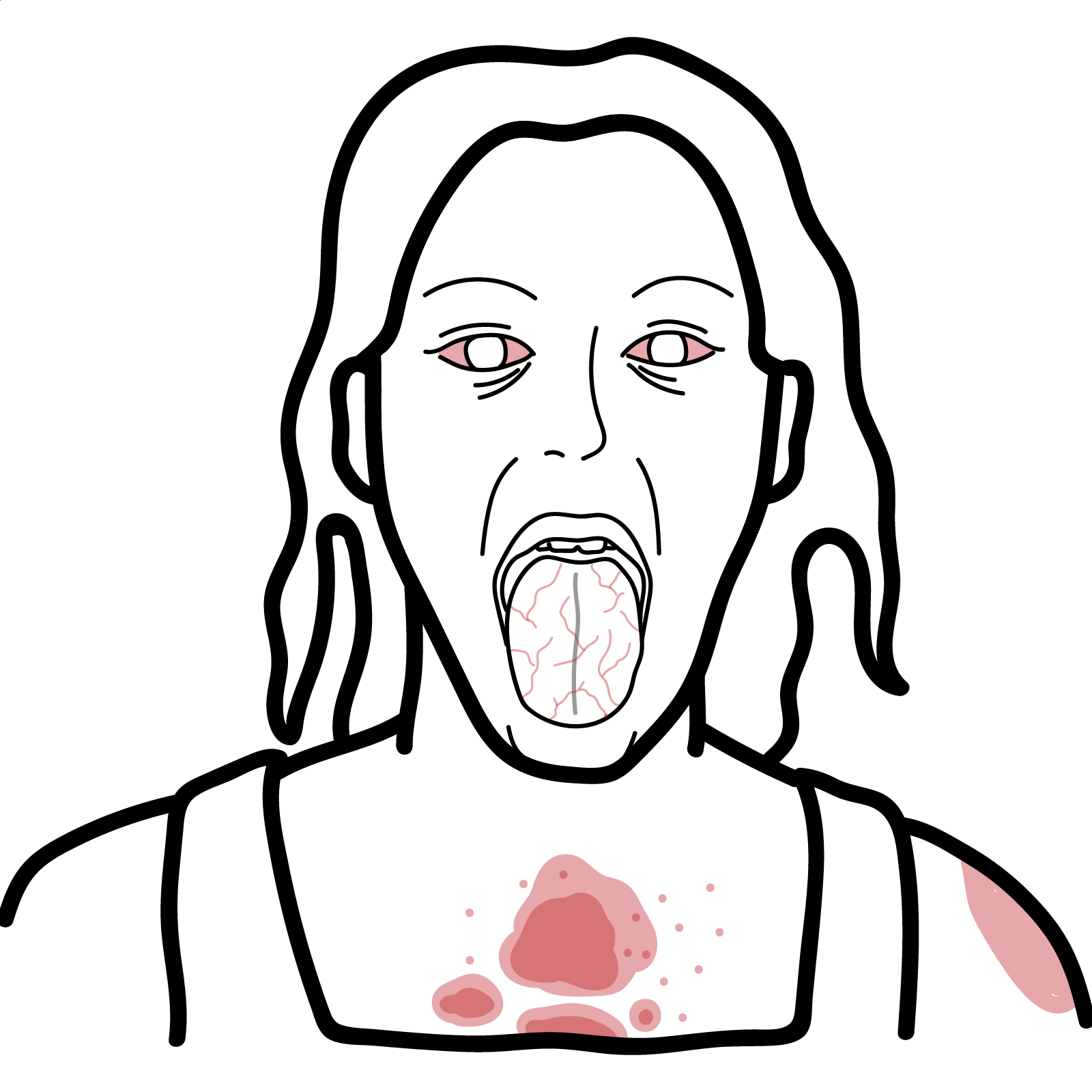
- Constitutional: fatigue, malaise, fever, weight loss, arthralgias
- Pulmonary: hemoptysis (may be absent), dyspnea, cough, chest pain (<50%), respiratory distress; crackles, cyanosis
- Renal: hematuria, proteinuria, oliguria, edema, hypertension, rapidly progressive renal failure (RPGN)
- Frequency: 60–80% dual involvement; 20–40% renal-only; <10% lung-only
Triad: hemoptysis + glomerulonephritis + anti-GBM antibodies — diagnostic triad of Goodpasture syndrome.
Remember
absence of hemoptysis does NOT rule out pulmonary involvement.
Diagnosis
- Anti-GBM antibody
- Renal biopsy: crescentic GN, linear IgG deposition on GBM by immunofluorescence
- ANCA (≈20–35% double positive)
- Complement levels; exclude other vasculitides or lupus
Differential diagnoses
- ANCA-associated vasculitis (e.g., GPA)—distinguished by presence of ANCA and pauci-immune GN
- SLE
- Other pulmonary hemorrhage causes (coagulopathy, infection)
Treatment
- Plasmapheresis: rapid removal of circulating anti-GBM antibodies
- High-dose corticosteroids plus cyclophosphamide or rituximab
- Azathioprine for maintenance
Supportive care: dialysis for renal failure; respiratory support or intubation for pulmonary hemorrhage
Complications and Prognosis
Complications
• Renal failure requiring long-term dialysis or transplant.
• Recurrent pulmonary hemorrhage.
• Infections due to immunosuppression.
• Rare relapse.
Prognosis:
• With early treatment: 5-year survival ~80%, >80% overall initial survival [8,4].
• Without treatment: nearly universally fatal.
• Poor prognostic factors: dialysis dependence at presentation, high creatinine, extensive crescents, delayed treatment
Remember
early intervention markedly improves outcomes.
References
- Lee JY, et al. Goodpasture Syndrome. MSD Manual Professional. 2023.
- Hellmark T, Segelmark M. Diagnosis and classification of Goodpasture’s disease. Div Nephrol. 2014.
- Li T, McAdoo SP. Anti-GBM disease. BMJ Best Practice. 2024.
- Kathuria P, Batuman V. Goodpasture Syndrome. Medscape. 2023.
- Longo L, Martellucci S, Fusconi M. Goodpasture’s syndrome: a clinical update. Autoimmun Rev. 2015.
- Wikipedia contributors. Goodpasture syndrome. Accessed 2025.




Discussion