Graves’ Disease


Graves’ disease is the most common cause of hyperthyroidism, caused by an autoimmune reaction in which TSH receptor autoantibodies stimulate the thyroid gland, leading to an increase in the production of thyroid hormones. It’s named after Dr Robert James Graves, an Irish physician who in 1835 gave one of the first thorough clinical descriptions linking goitre, palpitations and exophthalmos—the classic picture of autoimmune hyperthyroidism. The disease typically affects women more than men and primarily affects those between the ages of 30-50.
Goitre: diffuse enlargement of the thyroid from stimulatory autoantibodies.
Thyrotoxicosis: The clinical state of excess circulating thyroid hormone (T3/T4) from any source.Includes endogenous overproduction or passive release/exogenous intake.
Hyperthyroidism: a subset of thyrotoxicosis where the thyroid gland is overproducing hormone. All hyperthyroidism causes thyrotoxicosis, but not all thyrotoxicosis is hyperthyroidism.
Physiology of the thyroid gland
The hypothalamic-pituitary-thyroid axis plays a major role in regulating thyroid hormone levels in the body. These hormones play an integral role in metabolism and fetal development, all of which are governed by an intricate feedback mechanism in the HPT axis. Key structures in this axis include the hypothalamus, pituitary gland and thyroid, alongside the key hormones: thyrotropin-releasing hormone (TRH), thyroid-stimulating hormone (TSH) and thyroxine (T4) and triiodothyronine (T3).
The hypothalamus produces and secretes TRH, which then stimulates the cells of the anterior pituitary to produce TSH. TSH then stimulates the thyroid gland to produce and release T3 and T4, which are distributed throughout the body to regulate metabolism. The production of T3 and T4 creates a negative feedback loop where they inhibit the release of TRH and TSH once levels get too high. 1, 2
Being an autoimmune condition, Graves disease is more common in those with a positive family history. Environmental factors such as smoking, stress, infection and pregnancy are known to precipitate it. 3
Extrathyroidal manifestations (TSH-receptor expression outside the thyroid; immune-mediated fibroblast activation)
Graves disease most commonly presents with initial signs of hyperthyroidism first, these include:
Clinical examination
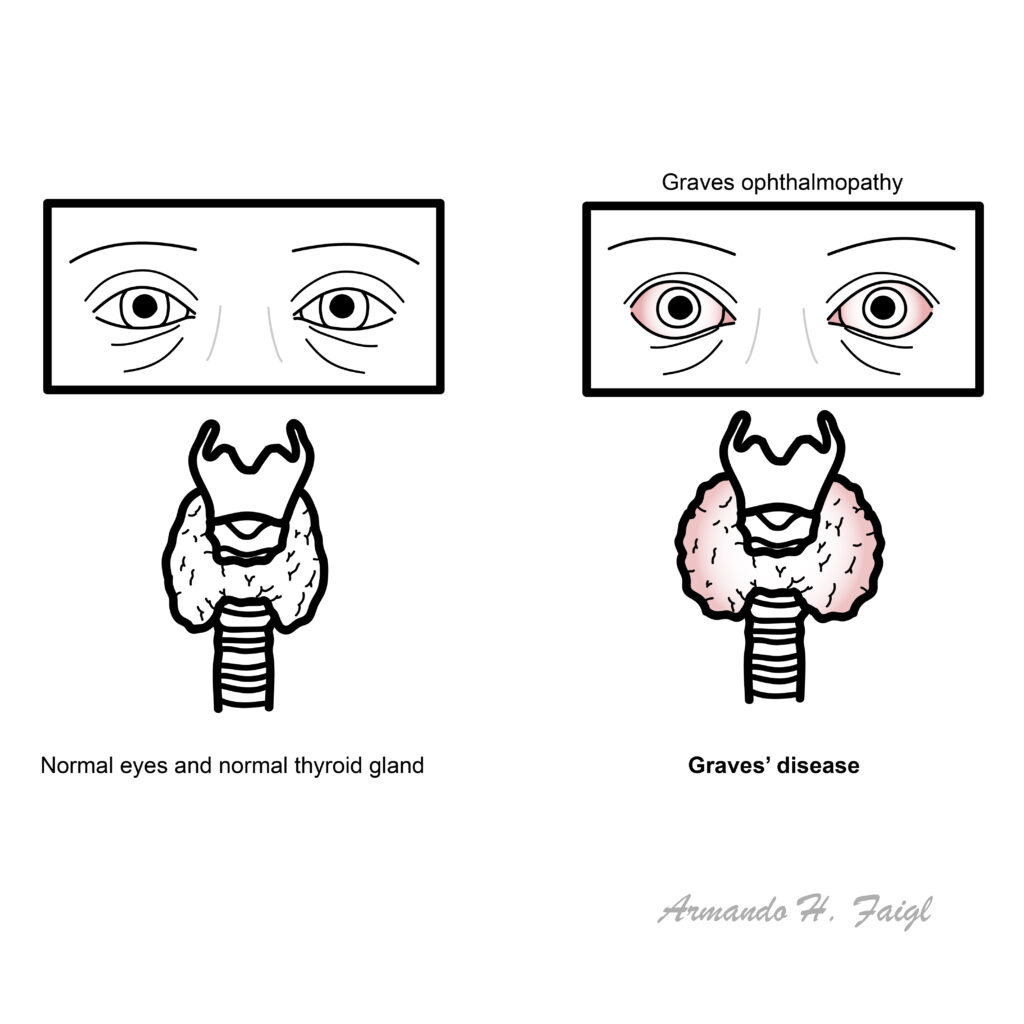
Graves ophthalmopathy features
Graves dermopathy features
Consider Graves’ disease in patients with new-onset atrial fibrillation, unexplained weight loss, or eye symptoms (like proptosis or lid retraction), even if classic hyperthyroid signs are absent—especially in the elderly.
Thyroid goitre: A goitre is a general term for any abnormal enlargement of the thyroid gland and is commonly seen in conditions which cause thyroid hyperplasia. It can be benign or malignant and is seen as a mass located in the neck that may or may not move with swallowing.
The diagnosis of Graves’ disease is made through both clinical evaluation and lab investigations.
Antithyroid peroxidase antibodies (anti-TPO) and thyroglobulin antibodies TgAbs) can be elevated in ALL forms of autoimmune thyroid disease and are thus not specific for Graves’.
Subclinical hyperthyroidism is a biochemical diagnosis (low TSH, normal T4/T3) with important treatment thresholds (especially TSH <0.1 mIU/L or high-risk patients).
Differential Diagnoses of hyperthyroidism
| Diagnosis | Definition | Clinical features |
| Graves disease | Autoimmune stimulation of TSH receptors by TRAb/TSI → diffuse overproduction | Diffuse goitre (± bruit/thrill), ophthalmopathy, dermopathy (pretibial myxoedema); hyperadrenergic symptoms; TRAb positive; diffuse high RAIU; Doppler shows markedly increased vascularity |
| Toxic multinodular goitre | Multiple autonomously functioning nodules producing excess hormone | Longstanding nodular goitre; typically older adults; atrial fibrillation common; patchy/heterogeneous RAIU; no eye disease |
| Toxic adenoma | Single autonomous “hot” nodule suppressing the rest of the gland | Solitary thyroid nodule; mild–moderate thyrotoxicosis; focal hot nodule with suppressed background on RAIU |
| hCG-mediated (gestational/trophoblastic) | Excess hCG stimulates TSH receptors | Early pregnancy; hyperemesis; mild goitre; low TSH, mildly ↑ FT4; often transient |
| Iodine-induced (Jod-Basedow) / Amiodarone type 1 | Iodine excess triggers overproduction in autonomous tissue or latent Graves | Occurs after contrast/amiodarone exposure; underlying nodular goitre common; RAIU often low/normal due to iodine saturation; Doppler shows increased flow in type 1 |
| TSH-secreting pituitary adenoma (TSHoma) | Pituitary tumour secreting inappropriately high TSH | High T4/T3 with non-suppressed or high TSH; goitre; headaches/visual field defects; pituitary MRI shows macroadenoma |
| Ectopic hormone production (e.g., struma ovarii) | Thyroid hormone synthesised outside the thyroid | Normal/small thyroid; persistent thyrotoxicosis; low thyroidal RAIU with extra-thyroidal uptake on whole-body scan |
| RAIU: Radioactive Iodine Uptake, TRAb: TSH receptor antibodies, TSI: thyroid-stimulating immunoglobulins | ||
The treatment of Graves’ disease is dependent on its presentation, but has two key aims:
before starting thionamide treatment, patients should be informed about side effects such as anaphylaxis, neutropenia and hepatotoxicity. In the first trimester of pregnancy, propylthiouracil should be used due to less teratogenic side effects.
Thionamides may be started prior to radioiodine therapy as it can transiently worsen symptoms.
Complications include:
Graves’ disease generally has a good prognosis if treated promptly. However, risk of relapse is common after antithyroid drug therapy. The disease has remission rates of approximately 45%, with most relapses occurring within 4 years of treatment. Risk factors of relapse include ophthalmopathy, young age, large goitre size and smoking.

Please confirm you want to block this member.
You will no longer be able to:
Please allow a few minutes for this process to complete.
Discussion